Thermo-tunable colorimetric detection of mercury(ii) ions driven by the temperature-dependent assembly and disassembly of a block copolymer†
Abstract
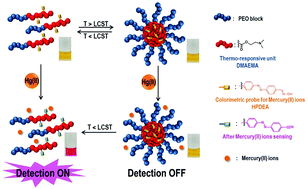
We report the synthesis of a thermo-responsive double-hydrophilic block copolymer (DHBC) and its tunable detection of mercury(II) ions modulated by a temperature-dependent morphological transition between unimers and micellar aggregates. 2-(Dimethylamino)ethyl methacrylate (DMAEMA) and (E)-2-((4-((4-formylphenyl)diazenyl)phenyl)(methyl)-amino) ethyl acrylate (FPDEA) were copolymerized from a poly(ethylene oxide) (PEO) macroinitiator via atom transfer radical polymerization (ATRP), resulting in the formation of a well-defined block copolymer (PEO-b-[p(DMAEMA-co-FPDEA)]). An oxime-containing polymeric probe, PEO-b-[p(DMAEMA-co-(E)-2-((4-((2-hydroxyimino phenyl)diazenyl)phenyl)(methyl)-amino) ethyl acrylate)] and PEO-b-[p(DMAEMA-co-HPDEA)], P1, comprising a permanently hydrophilic PEO block and a thermo-switchable p(DMAEMA-co-HPDEA) block was ultimately synthesized via post-polymerization modification using hydroxylamine, which caused the conversion of the aldehyde groups of PEO-b-[p(DMAEMA-co-FPDEA)] to aldoxime groups that are responsible for the colorimetric detection of mercury(II) ions. At solution temperatures below 55 °C, the value of the lower critical solution temperature (LCST) of the p(DMAEMA-co-HPDEA) block, P1 was present in water in the form of soluble unimers. P1 self-assembled to form multi-molecular micelles in water once the solution was heated above 55 °C. In this situation, no mercury(II) ion detection was observed, because mercury(II) ion-detecting HPDEA units are located in the hydrophobic core of the micelles, to which mercury(II) ions have no access. In the presence of mercury(II) ions, as the temperature of a P1 solution decreased below the value of the LCST of the p(DMAEMA-co-HPDEA) block, however, the said block became hydrophilic, resulting in the dissociation of micelles into unimers. Evidence indicated at this point that the turn-on detection of mercury(II) ions had been achieved, following the exposure of mercury(II) ion-detecting HPDEA units to the aqueous medium.


 Please wait while we load your content...
Please wait while we load your content...