Star polymers with acid-labile diacetal-based cores synthesized by aqueous RAFT polymerization for intracellular DNA delivery†
Abstract
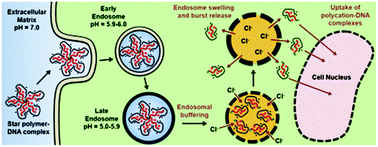
The application of DNA-based therapeutics holds much potential, but it is limited by the ability to successfully deliver and transfect target cells. Here, acid-labile diacetal-based star polymers were synthesized by a facile ‘arm-first’ heterogeneous reversible addition–fragmentation chain transfer polymerization in biologically compatible solvents, for use as DNA delivery vehicles. Their cytotoxicity, DNA binding and transfection rates were evaluated. The star polymer arms are based on the biocompatible oligo(ethylene glycol) methacrylate (OEGMA) monomer, the cationic 2-(dimethylamino)ethyl methacrylate (DMAEMA) monomer and their mixtures. Their cores were prepared by the acid-labile diacetal bis[(2-methacryloyloxy)ethoxymethyl] ether (MOEME) or the non-degradable ethylene glycol dimethacrylate (EGDMA) cross-linker, for comparison. The MOEME-based star polymers showed accelerated degradation in acidic conditions. The fully cationic star polymers, PDMAEMA26-MOEME4-star and PDMAEMA26-EGDMA6-star, showed higher cytotoxicity but also considerably better DNA complexation. The higher transfection efficiency obtained for PDMAEMA26-MOEME4-star (65.5%) compared to PDMAEMA26-EGDMA6-star (44.2%) can be attributed to the ability of the former to disassemble at endosomal pH, due to its acid-labile MOEME-based core. The ability of the star polymer–DNA complexes to escape the endo/lysosomal pathway was shown to be a key determinant of transfection efficiency, as assessed by confocal microscopy and flow cytometry. In conclusion, these star polymers show promise as biocompatible, pH-labile polymers for DNA delivery that can be synthesized without the need of toxic organic solvents.
- This article is part of the themed collection: Polymer Chemistry Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...