Light-responsive arylazopyrazole-based hydrogels: their applications as shape-memory materials, self-healing matrices and controlled drug release systems†
Abstract
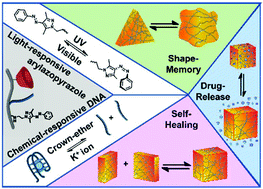
Carboxymethyl cellulose (CMC) chains functionalized only with β-cyclodextrin (β-CD), or with nucleic acids, β-CD and photoisomerizable arylazopyrazole (AAP), self-assemble into stimuli-responsive hydrogels which reveal stiffness-controlled properties. In the first system, β-CD modified CMC chains crosslinked by trans-bis-AAP (1) result in a hydrogel that can be switched between low-stiffness and high-stiffness states in response to alternating photoisomerization between trans-bis-AAP/cis-bis-AAP. In the second system, the CMC scaffold is modified with β-CD units, AAP and self-complementary nucleic acid (2) as tethers, leading to a high-stiffness hydrogel, cooperatively crosslinked by β-CD/trans-AAP and (2)/(2) duplexes. By alternating the photoisomerization of trans-AAP units to cis-AAP and back, the hydrogel is cycled between low-stiffness and high-stiffness states. In the third system, the use of G-quadruplexes as crosslinking motifs that respond to chemical triggers results in a hydrogel that reveals dual triggered stiffness properties, stimulated by light or K+-ions/crown-ether. The hybrid hydrogels are applied as light-responsive and dual triggered shape-memory, self-healing and controlled drug-release materials.


 Please wait while we load your content...
Please wait while we load your content...