A new healable polymer material based on ultrafast Diels–Alder ‘click’ chemistry using triazolinedione and fluorescent anthracyl derivatives: a mechanistic approach†
Abstract
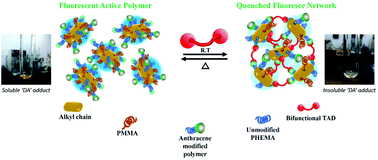
Since the last decade, the synthesis of self-healable polymers using Diels–Alder (DA) ‘click’ chemistry has been one of the major focus areas in the chemical community. Reversible covalent DA linkages lead to the synthesis of several materials which can be applicable from the biological to industrial fields. Herein, we report a new type of healable fluorescent polymer material based on an anthracene and 1,2,4-triazoline-3,5-dione (TAD) system which has several advantages over the conventional bismaleimide-based DA reaction. In this case, a reversible addition fragmentation chain-transfer (RAFT) polymerization technique was employed to prepare copolymers of 2-hydroxyethyl methacrylate (HEMA) and methyl methacrylate (MMA). The methacrylate copolymers bearing repeating pendant hydroxyl groups were coupled with anthracyl derivatives, where the aromatic groups acted as the ‘click’ partner to the TAD moiety. The addition of TAD derivatives to the anthracyl-modified copolymers led to the synthesis of an ultrafast cross-linked network under ambient conditions without any need of modular reagents. Rationalization of the ultrafast DA reaction mechanism of the TAD–anthracene system was carried out via density functional theory (DFT) calculations by taking different anthracene and TAD derivatives. Thermoreversible properties and healing characteristics were analyzed by DSC and microscopic analyses.


 Please wait while we load your content...
Please wait while we load your content...