Phosphazene superbase catalyzed ring-opening polymerization of cyclotetrasiloxane toward copolysiloxanes with high diphenyl siloxane content†
Abstract
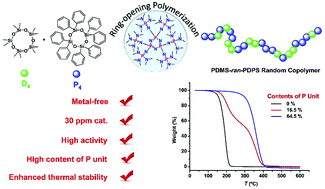
Polysiloxanes containing phenyl functional groups are a class of promising polymer materials with enhanced low-temperature flexibility, high-temperature stability and great damping properties. In this context, organic cyclic trimeric phosphazene bases (CTPBs) have been proved to be highly efficient for the ring-opening polymerization (ROP) of octamethylcyclotetrasiloxane (D4) and the copolymerization of D4 with octaphenylcyclotetrasiloxane (P4) under mild conditions. The polymerizations are fast and the resulting polymers have high molecular weights (Mn up to 1353 kg mol−1). The molecular structures of the obtained copolysiloxanes were characterized by 1H NMR and 29Si NMR, which demonstrate that as high as 64 mol% diphenyl siloxane units can be incorporated within the copolysiloxane chain. Remarkably, copolysiloxanes with different contents of the diphenyl siloxane group are found free of T-branches and Q-branches based on the 29Si NMR analysis, indicating a good controlled ROP by the current organocatalytic system. DSC analysis shows no crystallization exotherm or melting endotherm in the range of −70 °C to 50 °C for all the copolysiloxanes, confirming that incorporation of diphenyl groups successfully suppresses the undesirable crystallization of polydimethylsiloxane. Moreover, the thermal stability of copolysiloxanes was evaluated by TGA, showing dramatically enhanced thermal stability with the increment of the diphenyl siloxane content.


 Please wait while we load your content...
Please wait while we load your content...