Ferrocene-based (photo)redox polymerization under long wavelengths†
Abstract
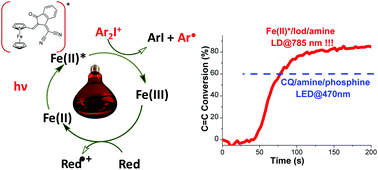
Ferrocene-based photoredox catalysis is proposed here for the first time. Aryl radicals generated from a Fe(II)*/Ar2I+ reaction can be used as initiating species for efficient free radical photopolymerization of methacrylate resins. Remarkably, these photoredox catalysts can also be used for redox free radical polymerization (without light) in combination with ammonium persulfate for unique access to dual cure (photochemical/thermal redox) systems. The addition of a third component (amine, phosphine or vitamin C reducing agents) enables the regeneration of the catalysts and greatly enhances the radical generation. The motivation with these dual cure systems is to develop orthogonal chemistries where a latent redox polymerization (without light) is able to cure any thickness of polymers (or composite) in combination with fast photopolymerization processes in the irradiated areas. Chemical mechanisms will be discussed in detail using cyclic voltammetry, electron spin resonance spin trapping (ESR-ST), UV-vis-NIR spectroscopy, free energy calculations and molecular modeling at the density functional theory (DFT) level. This study represents, to the best of our knowledge, the first photochemically active iron catalysts that are also efficient in thermal redox catalysis.


 Please wait while we load your content...
Please wait while we load your content...