Salen complexes of zirconium and hafnium: synthesis, structural characterization and polymerization studies†
Abstract
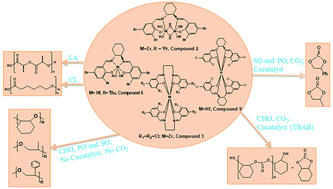
A complete library of Zr and Hf complexes bearing salen ligands were synthesized and characterized by various spectroscopic techniques and X-ray crystallography. The stoichiometry of the reactions for the preparation of these complexes alters on varying the ligands and leads to different products. For Zr we observe mononuclear and dinuclear complexes. In the dinuclear complexes, each Zr centre adopts a distorted octahedral geometry. The metal centers are bridged with two isopropoxide groups and each metal center has two terminal isopropoxide moieties attached to it. The mononuclear Zr complex adopts a distorted square antiprism geometry. Hf complexes adopt both mononuclear distorted octahedral and distorted square antiprism geometries. The catalytic activity towards the ring-opening polymerization (ROP) of lactide (LA) and ε-caprolactone (ε-CL) was assessed. These compounds were found to be potent catalysts, resulting in the formation of products with very high number average molecular weight (Mn) values with controlled dispersity (Đ). All the complexes were found to be highly active towards the copolymerization of cyclohexene oxide (CHO) with CO2 generating polycarbonates showing good turnover frequencies (TOF). In the cases of styrene oxide (SO) and propylene oxide (PO), the formation of cyclic styrene carbonate and cyclic propylene carbonate, respectively, is witnessed. The steric and electronic nature of the substituents of ancillary ligands influences the catalytic activity of these complexes, with the electron donating substituent reducing the activity. The kinetics of L-LA, rac-LA and ε-CL polymerization was studied in detail and revealed that the polymerization follows first-order kinetics. The mechanisms of respective polymerization processes were also investigated. Analysis of the MALDI-TOF and 1H NMR spectra of a low Mn oligomer from rac-LA polymerization reveals that the isopropoxide group initiates the ROP.


 Please wait while we load your content...
Please wait while we load your content...