Structure and luminescence properties of supramolecular polymers of amphiphilic aromatic thioether–peptide conjugates in water†
Abstract
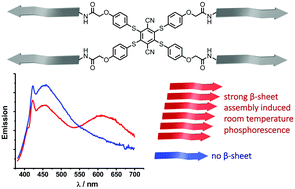
We present the preparation of luminophore–peptide conjugates that self-assemble into supramolecular polymers in neutral buffer. To this end, we have prepared a small library of six conjugates with varying substitution patterns of the aromatic thioethers, as well as varying amino acid sequences. The latter have allowed us to tune the thermodynamic driving force for self-assembly and probe their photoluminescent properties either in the monomeric or polymeric state, while fully avoiding selective solvent techniques or organic solvent mixtures. All of the supramolecular structures were characterised with transmission electron microscopy, circular dichroism measurements, as well as steady-state and time-resolved photoluminescence spectroscopy. In the case of the tetra-substituted (phenylthio)phthalonitrile luminophore we observe a long-lived, red-shifted emission at room temperature which we attribute to phosphorescence upon self-assembly into anisotropic supramolecular polymers. Such an aggregation induced phosphorescence at wavelengths above 600 nm is very promising for the development of tuneable luminophores for applications of multifunctional supramolecular materials in biological systems and time-gated optical microscopy.
- This article is part of the themed collection: Pioneering Investigators


 Please wait while we load your content...
Please wait while we load your content...