Enzymatic synthesis of unsaturated polyesters: functionalization and reversibility of the aza-Michael addition of pendants†
Abstract
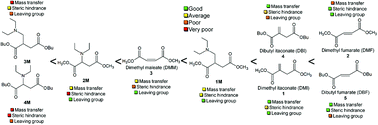
With society's increasing awareness of the problems caused by plastic pollution, the switch to renewable, biodegradable materials is nowadays a must for several producers and consumers. In the present work, the biocatalyzed synthesis of fully renewable polyesters containing lateral vinyl functionalities derived from itaconic, fumaric and maleic acid is presented. Here, an environmentally friendly synthesis was therefore combined with the possibility of obtaining materials that could be functionalized via post-polymerization modifications such as the aza-Michael additions of diethylamine. Moreover, the possibility of reversing the polymer's post-functionalization via a temperature-driven process to obtain free lateral vinyl groups again is also described and analyzed for several polymers based on itaconates and 1,4-butanediol, 1,6-hexanediol and 1,8-octanediol. The collected results set the basis for the development of switchable polymers where pharmaceuticals or other chemicals/polymers can be coupled to the polymeric backbone and released upon changing the material's conditions.


 Please wait while we load your content...
Please wait while we load your content...