Quantification of micropolarity and microviscosity of aggregation and salt-induced gelation of sodium deoxycholate (NaDC) using Nile red fluorescence†
Abstract
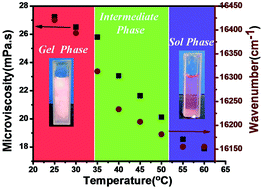
This study reports the utility of the hydrophobic probe Nile Red (NR) to understand the concentration induced microenvironmental changes of sodium deoxycholate (NaDC) bile salt from the premicellar to postmicellar range. The spectroscopic properties like absorbance value, fluorescence intensity and fluorescence lifetime of NR are significantly sensitive towards different states of aggregation of NaDC bile salt. The critical aggregation concentrations of different states (dimer to primary micellar aggregates (1.0 mM), secondary micellar aggregates (7.0 mM), and higher micellar aggregates (14 mM)) have been determined from the absorbance value and fluorescence intensity measurements. The ET(30) polarity parameter values suggest a considerable decrease in the micropolarity with an increase in NaDC concentrations. Furthermore, the spectroscopic properties of NR are also sensitive towards the NaCl induced gelation process of NaDC bile salt. Changes in the micropolarity and microviscosity of the NaDC + NaCl mixed system have been estimated using the emission maximum value (cm−1) and fluorescence lifetime values of NR with an increase in the NaCl concentration. Microviscosity of the medium increases from ∼19 mPa s to ∼26 mPa s from the sol phase to the gel phase. Temperature dependence of both size and phase changes of the NaDC + NaCl (30 mM + 1 M) gel network has been studied using differential scanning calorimetry and dynamic light scattering studies. Temperature induced polarity and microviscosity changes of the NaDC + NaCl (30 mM + 1 M) gel network have also been studied using the fluorescence properties of NR.


 Please wait while we load your content...
Please wait while we load your content...