Ultrafast excited state dynamics of nonfluorescent cyclopheophorbide-a enol, a catabolite of chlorophyll-a detoxified in algae-feeding aquatic microbes†
Abstract
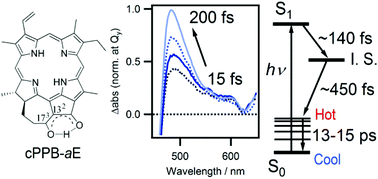
For photosynthetic organisms that nourish the earth's biosphere, chlorophylls (Chls) are the major pigments utilized for light harvesting and primary charge separation. Although Chl molecules are effective photosensitizers, they are inevitably phototoxic to living organisms due to the facile generation of highly oxidative singlet oxygen (1O2) through triplet energy transfer from their photoexcited states to oxygen molecules. Such phototoxicity of Chls is a major problem for translucent microbes that feed on photosynthetic algae. Recently, it has been reported that the metabolic conversion of Chls-a/b to 132,173-cyclopheophorbide-a/b enols (cPPB-a/bEs) is the detoxification mechanism for algivorous protists. cPPB-a/bEs are colored π-conjugated cyclic tetrapyrroles but are nonfluorescent due to efficient nonradiative decay. In this study, femtosecond time-resolved transient absorption spectroscopy was applied to cPPB-aE with the aim of understanding its quenching mechanism. As a result, we have captured the ultrafast generation of an intermediate state (∼140 fs) that leads to the rapid internal conversion to the ground state (∼450 fs).


 Please wait while we load your content...
Please wait while we load your content...