Total syntheses of seminolipid and its analogues by using 2,6-bis(trifluoromethyl)phenylboronic acid as protective reagent†
Abstract
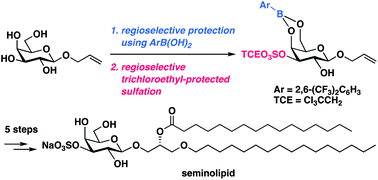
A concise total synthesis of seminolipid, a sulfoglycolipid, has been achieved; key features include regioselective, tin-free sulfation of allyl β-D-galactopyranoside using 2,6-bis(trifluoromethyl)phenylboronic acid as protective reagent, stereoselective epoxidation, and site-selective acylation. The utility of this divergent synthetic approach to introduce 2,2,2-trichloroethyl-protected sulfate group at an early stage without toxic and environmentally unfavorable tin reagents was demonstrated by the syntheses of three seminolipid analogues with different side-chains from the common intermediate.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...