1,2-Addition versus homoconjugate addition reactions of indoles and electron-rich arenes to α-cyclopropyl N-acyliminium ions: synthetic and computational studies†
Abstract
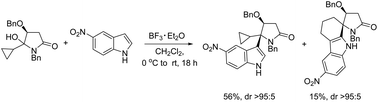
An investigation of the reactivity of α-cyclopropyl N-acyliminium ions towards indoles has resulted in the unprecedented synthesis of 5-cyclopropyl-5-(3-indoyl)pyrrolidin-2-ones via 1,2-addition reactions and, in the case of highly electron deficient indoles and electron rich arenes, spiroheterocycles via sequential homoconjugate and 1,2-addition reactions with often high diastereoselective control at the C-5 quaternary stereocentres. Computational studies provided support for the proposed mechanisms and stereochemical outcome of these reactions, clearly showing that the 1,2-addition pathway is kinetically controlled. In reactions where the 1,2-adduct is destabilised, for example when the arene ring is less nucleophilic, the 1,2-addition is reversible and the thermodynamically preferred homoconjugate addition and subsequent rearrangement and cyclisation reactions become the major pathway.
- This article is part of the themed collections: Synthetic methodology in OBC and Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...