Recent advances in the direct functionalization of quinoxalin-2(1H)-ones
Abstract
The direct C3-functionalization of quinoxalin-2(1H)-ones via C–H bond activation has recently attracted considerable attention, due to their diverse biological activities and chemical properties. This review will focus on the recent achievements, mainly including arylation, alkylation, acylation, alkoxycarbonylation, amination, amidation and phosphonation of quinoxalin-2(1H)-ones. Their mechanisms are also discussed.
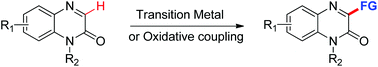
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...