Catalytic asymmetric cycloaddition reactions of enoldiazo compounds
Abstract
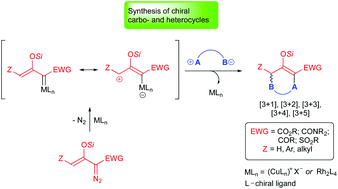
This review describes catalytic asymmetric cycloaddition reactions of silyl-protected enoldiazo compounds for the construction of highly functionalized carbo- and heterocycles which possess one or more chiral center(s). The enoldiazo compound or its derivative, donor–acceptor cyclopropene, form electrophilic vinylogous metal carbene intermediates that combine stepwise with nucleophilic dipolar reactants to form products from [3 + 1]-, [3 + 2]-, [3 + 3]-, [3 + 4]-, and [3 + 5]-cycloaddition, generally in high yield and with exceptional stereocontrol and regioselectivity.
- This article is part of the themed collections: Catalysis & biocatalysis in OBC and Carbenes in Organic Synthesis


 Please wait while we load your content...
Please wait while we load your content...