Highly efficient N2 fixation catalysts: transition-metal carbides M2C (MXenes)†
Abstract
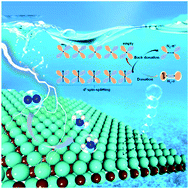
The development of highly efficient metal or metal compound electrocatalysts under mild conditions has always been a challenging task for N2 reduction. Herein, we show that pristine two-dimensional (2D) MXenes are promising N2 electroreduction catalysts due in part to the availability of multiple active sites per unit area. We systematically explore a series of 3d, 4d and 5d-transition metal M2C (M = Sc, Ti, V, Cr, Mn, Fe, Zr, Nb, Mo, Ta and Hf) MXenes and compute their limiting potentials for the N2 reduction reaction (NRR). We find that 4d4-Mo2C gives rise to the lowest free-energy barrier (ΔG) of 0.46 eV, among the synthesized M2C MXenes as of today. More importantly, we find that two hypothetical MXenes, 3d5-Mn2C and 3d6-Fe2C, possess even lower ΔG of 0.28 and 0.23 eV, respectively, compared to the state-of-the-art 4d4-Mo2C, thereby likely being more efficient NRR catalysts. The N2 capture strength, a key parameter of the potential-limiting step, is found to be closely related to the d-electron arrangement on the occupied and empty spin-split d-orbitals. Hence, the excellent NRR performance of Mn2C and Fe2C can be attributed to the desirable half-filled 3d5 or 3d6 electron arrangements. The adsorption of N2 on Mn2C results in the donation of 1σ electrons to the empty spin-down 3d orbitals of Mn. The donated electrons weaken the N2 adsorption strength and lower the energy barrier of the potential-limiting step of hydrogenation. The insights obtained from this comprehensive study offer guidance to design new and efficient electrocatalysts for N2 fixation.

 Please wait while we load your content...
Please wait while we load your content...