Influence of the solvent environment on luminescent centers within carbon dots†
Abstract
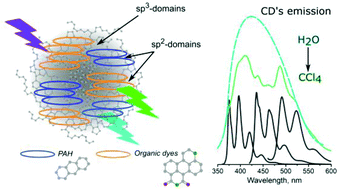
Carbon dots (CDs) are luminescent nanomaterials, with potential use in bioimaging and sensorics. Here, the influence of the surrounding solvent media on the optical properties of CDs synthesized from the most commonly employed precursors, namely citric acid and ethylenediamine, is investigated. The position of optical transitions of CDs can be tuned by the change of pH and solvent polarity. The most striking observation is related to the interaction of CDs with chlorine containing solvents, which results in resolving a set of narrow peaks within both the absorption and PL bands, similar to those observed for polycyclic aromatic hydrocarbons or organic dyes. We assume that the chlorine containing molecules penetrate the surface layers of CDs, which results in an increase of the distance between the luminescent centers; this correlates well with an enhanced D-band in their Raman spectra. A model of CDs composed of a matrix of hydrogenated amorphous carbon with the inclusions of sp2-domains formed by polycyclic aromatic hydrocarbons and their derivatives is suggested; the latter are stacked ensembles of the luminophores and are considered as the origin of the emission of CDs.

 Please wait while we load your content...
Please wait while we load your content...