N,S–Codoped hierarchical porous carbon spheres embedded with cobalt nanoparticles as efficient bifunctional oxygen electrocatalysts for rechargeable zinc-air batteries†
Abstract
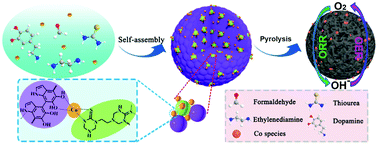
Rational design and fabrication of cost-effective, efficient bifunctional electrocatalysts is fundamentally important for the air cathode of metal–air batteries. Herein, a Co(II) ion-driven self-assembly strategy is described for the synthesis of cobalt-based nanostructured transition metal compounds (Co-NTMCs) embedded in nitrogen and sulfur codoped hierarchical porous carbon submicrospheres (Co-NTMCs@NSC), where condensation of thiourea-ethylenediamine-formaldehyde resin (TEFR) is induced by Co(II) ions which is simultaneously assembled with polydopamine to form a multifunctional precursor through coordinated interaction. The resulting Co-NTMCs@NSC sample comprises abundant hierarchical porous textures, a high content of active cobalt species including the nanoparticles of Co, Co3O4 and amorphous CoSx, and a considerable amount of defective structures. These characteristics lead to remarkable oxygen electrocatalytic activities, with a half-wave potential of +0.833 V vs. RHE, which is comparable to that of commercial Pt/C for the oxygen reduction reaction (ORR), and a lower overpotential of 284 mV than RuO2 at 10 mA·cm−2 for the oxygen evolution reaction (OER) in alkaline media. Furthermore, its operational stability is also much higher than that of commercial RuO2 and Pt/C catalysts. When used as a breathing air electrode for Zn–air batteries, Co-NTMCs@NSC shows a higher open-circuit voltage (1.509 V), higher discharge power density (262 mW cm−2) and better charge–discharge reversibility than commercial Pt/C. The results from the present work suggest that controlled assembly of functional polymers may be exploited for the preparation of doped carbon/metal nanoparticle nanocomposites as viable, high-performance catalysts for electrochemical energy technologies.


 Please wait while we load your content...
Please wait while we load your content...