Strong pyrrolic-N–Pd interactions boost the electrocatalytic hydrodechlorination reaction on palladium nanoparticles†
Abstract
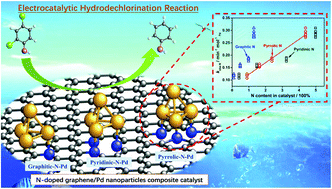
We demonstrated that heteroatomic nitrogen (N) doping of graphene can significantly enhance the performance of the graphene–palladium nanoparticle composite catalyst (N/G-Pd) in the electrocatalytic hydrodechlorination (EHDC) reaction. Specifically at −0.80 V (vs. Ag/AgCl), the N/G-430-Pd (prepared at 430 °C, pyridinic/pyrrolic-N-rich) and N/G-900-Pd (prepared at 900 °C, pyridinic/graphitic-N-rich) with equivalent total N content delivered the apparent rate constants (kobs) of 0.28 and 0.20 min−1 molPd−1 in removing 2,4-dichlorophenol, much higher than the 0.13 min−1 molPd−1 of the C-Pd. Additionally, we identified the determinant role of pyrrolic-N in boosting EHDC from the linear relationship between kobs-N and the pyrrolic-N content in the catalyst. Combined experimental and DFT analyses revealed that the positive effect of N doping originated from the strong N–Pd interactions, which modulated the Pd electronic structure and its interactions with the reactant and EHDC products (phenol and Cl−). The pyrrolic N–Pd bond was favorable as it could balance the reactant adsorption and the product desorption.

 Please wait while we load your content...
Please wait while we load your content...