Elucidating ligand effects in thiolate-protected metal clusters using Au24Pt(TBBT)18 as a model cluster†
Abstract
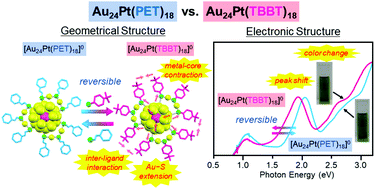
2-Phenylethanethiolate (PET) and 4-tert-butylbenzenethiolate (TBBT) are the most frequently used ligands in the study of thiolate (SR)-protected metal clusters. However, the effect of difference in the functional group between these ligands on the fundamental properties of the clusters has not been clarified. We synthesized [Au24Pt(TBBT)18]0, which has the same number of metal atoms, number of ligands, and framework structure as [Au24Pt(PET)18]0, by replacing ligands of [Au24Pt(PET)18]0 with TBBT. It was found that this ligand exchange is reversible unlike the case of other metal-core clusters. A comparison of the geometrical/electronic structure and stability of the clusters between [Au24Pt(PET)18]0 and [Au24Pt(TBBT)18]0 revealed three things with regard to the effect of ligand change from PET to TBBT on [Au24Pt(SR)18]0: (1) the induction of metal-core contraction and Au–S bond elongation, (2) no substantial effect on the HOMO–LUMO gap but a clear difference in optical absorption in the visible region, and (3) the decrease of stabilities against degradation in solution and under laser irradiation. By using these two clusters as model clusters, it is expected that the effects of the structural difference of ligand functional-groups on the physical properties and functions of clusters, such as catalytic ability and photoluminescence, would be clarified.


 Please wait while we load your content...
Please wait while we load your content...