Magnet-activatable nanoliposomes as intracellular bubble microreactors to enhance drug delivery efficacy and burst cancer cells†
Abstract
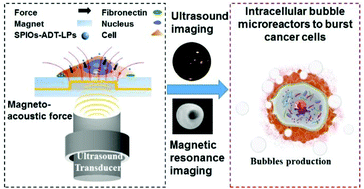
To address the thereapeutic challenges in clinical cancer treatment and guarantee efficient and rapid intracellular delivery of drugs while evading efflux and chemotherapy resistance, herein, we designed a liposomal nanostructure equipped with superparamagnetic iron oxide nanoparticles (SPIOs) and anethole trithione (ADT, a hydrogen sulfide (H2S) donor drug). At first, by spatially focused manipulation of the external static magnetic field (SMF), the SPIOs and ADT-loaded liposomes (SPIOs-ADT-LPs) could rapidly overcome the cell membrane barrier to enter the cytoplasm, which could be imaged by magnetic resonance imaging (MRI). Sequentially, the intracellular release of ADT drugs was triggered by enzymatic catalysis to generate acoustic-sensitive H2S gas. At the beginning, during the production of H2S at low concentrations, the cell membrane could be permeabilized to further increase the cellular uptake of SPIOs-ADT-LPs. The continued generation of H2S gas bubbles, imaged by ultrasound (US) imaging, further enhanced the intracellular hydrostatic pressure (above 320 pN per cell) to physically unfold the cytoskeleton, leading to complete cell death. The magneto-acoustic approach based on SPIO-ADT-LPs as intracellular bubble reactors leads to improved anticancer cell efficacy and has potential applications for novel MRI/US dual image-guided bubble bursting of cancer cells.
- This article is part of the themed collection: International Year of the Periodic Table: Applications for magnetic materials


 Please wait while we load your content...
Please wait while we load your content...