Bone formation promoted by bone morphogenetic protein-2 plasmid-loaded porous silica nanoparticles with the involvement of autophagy†
Abstract
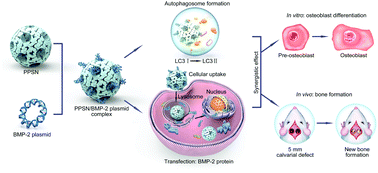
Gene therapy is one of the most common and effective ways for the regeneration of defective bone tissue, but even highly efficient gene delivery vectors are insufficient. In this study, bone morphogenetic protein-2 plasmid (pBMP-2) was encapsulated by polyethylenimine-modified porous silica nanoparticles (PPSNs), which were synthesized via an ethyl ether emulsion method. Owing to the high specific surface area and high absorption characteristics, low cytotoxicy PPSNs can efficiently load and protect pBMP-2. The resulting PPSN/pBMP-2 can transfect MC3T3-E1 cells effectively to promote osteogenic differentiation and increase calcium deposition in vitro. Interestingly, the mass of calcium deposition nodules decreased dur to the presence of an autophagy inhibitor, demonstrating that PPSNs stimulated the autophagy pathway. Because of their excellent biocompatibility, high transfection efficiency, and ability to stimulate autophagy, the as-prepared PPSN/pBMP-2 could efficiently transfect local cells in a defect area in vivo. Micro-computed tomography and histological images demonstrated that PPSN/pBMP-2 could efficiently promote new bone formation in a 5 mm sized rat calvarial defect model. Taken together, our newly synthesized PPSNs could efficiently carry pBMP-2 and deliver it to the target cells as well as stimulating the autophagy pathway, resulting in significant osteogenic differentiation and bone regeneration.


 Please wait while we load your content...
Please wait while we load your content...