Immobilisation of β-galactosidase within a lipid sponge phase: structure, stability and kinetics characterisation†
Abstract
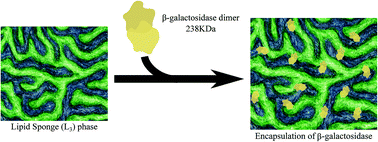
In the formulation of an active enzyme enclosed in a matrix for controlled delivery, it is a challenge to achieve a high protein load and to ensure high activity of the protein. For the first time to our knowledge, we report the use of a highly swollen lipid sponge (L3) phase for encapsulation of the large active enzyme, β-galactosidase (β-gal, 238 kDa). This enzyme has large relevance for applications in, e.g. the production of lactose free milk products. The formulation consisted of diglycerol monooleate (DGMO), and a mixture of mono-, di- and triglycerides (Capmul GMO-50) stabilised by polysorbate 80 (P80). The advantage of this type of matrix is that it can be produced on a large scale with a fairly simple and mild process as the system is in practice self-dispersing, yet it has a well-defined internal nano-structure. Minor effects on the sponge phase structure due to the inclusion of the enzyme were observed using small angle X-ray scattering (SAXS). The effect of encapsulation on the enzymatic activity and kinetic characteristics of β-galactosidase activity was also investigated and can be related to the enzyme stability and confinement within the lipid matrix. The encapsulated β-galactosidase maintained its activity for a significantly longer time when compared to the free solution at the same temperature. Differences in the particle size and charge of sponge-like nanoparticles (L3-NPs) with and without the enzyme were analysed by dynamic light scattering (DLS) and zeta-potential measurements. Moreover, all the initial β-galactosidase was encapsulated within L3-NPs as revealed by size exclusion chromatography.


 Please wait while we load your content...
Please wait while we load your content...