Understanding the mechanism of metal-induced degradation in perovskite nanocrystals†
Abstract
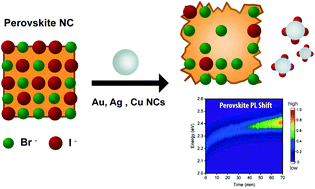
A deeper understanding of the perovskite–metal chemistry is crucial to elucidate the instability problems at the device level that can be caused by such interactions. Here, we study the reactions occurring between CsPbX3 (X = Br, BrI, I) perovskite and metal (M = Ag, Cu, Au) nanocrystals. We demonstrate a fast (<1 hour) optical and structural degradation of the I-containing nanocrystals driven by the formation of metal iodides with reaction kinetics according to the following order Cu < Ag < Au. These results point to the need for thoughtful considerations while constructing optoelectronic devices out of all-inorganic CsPbX3 nanocrystals, where the use of contact metals is a necessity.
- This article is part of the themed collection: Nanoscale 10th Anniversary Special Issue


 Please wait while we load your content...
Please wait while we load your content...