Exploring the chelation-based plant strategy for iron oxide nanoparticle uptake in garden cress (Lepidium sativum) using magnetic particle spectrometry†
Abstract
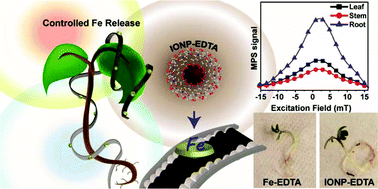
Although iron is one of Earth's most abundant elements, its availability to plants remains an agricultural challenge, particularly in high pH environments. At high pH, iron forms insoluble ferric oxide–hydroxides that makes it inaccessible to plants. It is estimated that 30% of the world's cropland is too alkaline for optimal plant growth. Staple crops, like rice, are particularly susceptible to iron deficiency, thereby, necessitating the need for continued research in developing iron-based fertilizers. Recent studies have demonstrated the potential of using iron oxide nanoparticles (IONPs) as fertilizers to address iron deficiency in plants, but some studies have generated conflicting results. One of the major challenges associated in investigating IONP plant uptake and translocation is the inability to distinguish between intact IONPs versus leached iron ions. In this study, we utilized a new approach based on magnetic particle spectrometry (MPS) to monitor the uptake and distribution of different sized (10 and 20 nm) chelated IONPs in plants. We exposed garden cress (Lepidium sativum) plants to EDTA-capped IONPs and observed an 8-fold enhancement in total biomass and 1.4 times increase in chlorophyll production compared to plants treated with a commercial chelated iron fertilizer (Fe-EDTA). Moreover, we demonstrated that the uptake and tissue distribution of IONPs can be quantitatively monitored using MPS, and the results of the analysis were validated by atomic absorption spectroscopy, which is the conventional method used to study IONP plant uptake. Our study demonstrates that MPS is a reliable, sensitive, and effective analytical tool for the development of IONP-based fertilizers.


 Please wait while we load your content...
Please wait while we load your content...