Ultrafine CoO nanoparticles as an efficient cocatalyst for enhanced photocatalytic hydrogen evolution†
Abstract
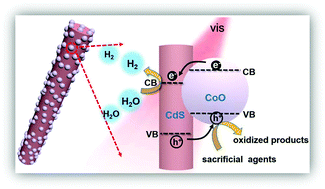
In order to further enhance the performance of photocatalysts, cocatalysts are used to accelerate the photocatalytic reactions. Herein, ultrafine cobalt oxide (CoO) nanoparticles are synthesized through a novel bottom-up strategy and explored as an efficient non-noble cocatalyst to dramatically promote the photocatalytic hydrogen evolution rate of CdS nanorods. CdS/CoO heterostructures, consisting of highly dispersed 3–5 nm CoO nanoparticles anchored on the CdS nanorods, can provide a high photocatalytic hydrogen evolution rate of 6.45 mmol g−1 h−1 (∼36 times higher than that of bare CdS nanorods) in the visible-light region (>420 nm). Combined X-ray photoelectron spectroscopy and X-ray absorption near edge spectroscopy analyses suggest Co–S bond formation between CoO and CdS, which guarantees efficient migration and separation of photogenerated charge carriers. This work provides a new avenue for adopting CoO as an effective cocatalyst for enhanced photocatalytic hydrogen production in the visible-light region.


 Please wait while we load your content...
Please wait while we load your content...