Facile in situ synthesis of ultrasmall near-infrared-emitting gold glyconanoparticles with enhanced cellular uptake and tumor targeting†
Abstract
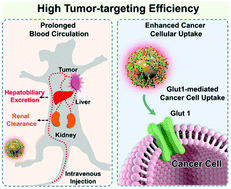
The simultaneous possession of high tumor-targeting efficiency, long blood circulation, and low normal-tissue retention is critical for future clinically translatable nanomedicines. Herein, we reported a facile in situ glycoconjugation strategy for the synthesis of near-infrared (NIR)-emitting gold glyconanoparticles (AuGNPs, ∼2.4 nm) using 1-thio-β-D-glucose as both the surface ligand and the reducing agent in the presence of a gold precursor. The ultrasmall AuGNPs showed similar low healthy organ retention to that of the renal-clearable ultrasmall nonglyconanoparticles, but ∼10 and 2.5 times higher in vitro and in vivo tumor-targeting efficiencies, respectively, were observed. This facile glycoconjugation strategy of ultrasmall AuGNPs was found to show activity towards glucose transporters in the cancer cells and prolonged blood circulation with both renal and hepatobiliary clearance pathways, which synergistically enhanced the tumor targeting of the ultrasmall AuGNPs. This discovery provides a smart strategy for the improvement in tumor targeting by ultrasmall NPs and further strengthens our understanding of glycoconjugation in designing future clinically translatable nanomedicines.
- This article is part of the themed collection: 2019 Nanoscale HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...