Transition from vesicles to nanofibres in the enzymatic self-assemblies of an amphiphilic peptide as an antitumour drug carrier†
Abstract
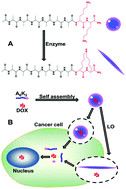
Amphiphilic peptides modified by molecular design can self-assemble into specific nanostructures with interesting applications in the fields of biomedicine and biotechnology. Lysyl oxidase (LO) is ubiquitous in human serum. However, enzymatic self-assembly of amphiphilic peptides remains a challenge for lipid-soluble drug delivery under the induction of LO. Here, we designed a positively charged amphiphilic peptide, A6K2, that could stably self-assemble to form nanovesicles. The lysine in the peptide molecule could be covalently cross-linked under enzyme catalysis, and the major transition was from random coil to β-sheet secondary structures, eventually leading to the destruction of the peptide nanovesicles. The lipid-soluble antitumour drug doxorubicin (DOX) as a model drug could be loaded into the hydrophobic core of the nanovesicles formed by the amphiphilic peptide A6K2, even though DOX was not covalently linked to the peptide monomer. The amount of DOX-encapsulated A6K2 nanovesicles in human hepatocellular carcinoma BEL-7402 cells was significantly higher than that in human liver L02 cells, indicating excellent selectivity. The amphiphilic peptide A6K2 inhibited tumour cell growth and had low cytotoxicity to mammalian cells, and it showed antibacterial activity against G+ and G− bacteria. These advantages make enzymatic self-assembling A6K2 nanovesicles of great interest in drug delivery for biomedical applications.


 Please wait while we load your content...
Please wait while we load your content...