Dynamics of carbohydrate strands in water and interactions with clay minerals: influence of pH, surface chemistry, and electrolytes†
Abstract
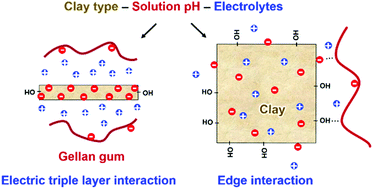
Carbohydrate hydrogels are extensively used in pharmaceuticals and engineered biomaterials. Molecular conformations, assembly, and interactions of the carbohydrate strands with stabilizers such as clay minerals in aqueous solution are difficult to quantify in experiments and the hydrogel properties remain largely a result of trial-and-error studies. We analyzed the assembly of gellan gum in aqueous solution and interactions with dispersed clay minerals in all-atomic detail using molecular dynamics simulation, atomic force microscopy (AFM), and comparisons to earlier measurements. Gellan strands associate at low pH values of 2 and gradually disassemble to double strands with weak association of −0.4 kcal per mole carbohydrate ring as the pH values increases to 9. Ionization of the carbonic acid side groups in the backbone extends the chains and accelerates the conformational dynamics via rapidly changing intramolecular ion bridges. Gellan interactions with clay minerals depend on the strength of electric triple layers between clay, cations, and anionic polymer strands, as well as weaker hydrogen bonds along the edges, which are tunable as a function of the clay surface chemistry, local ionic strength, and pH values. Interaction energies range from −4 to +6 kcal per mol carbohydrate ring and were most favorable for electric triple layers with high charge mobility, which can be achieved for intermediate cation exchange capacity of the clay mineral and high pH values to increase ionization of the clay edges and of the polymer. The findings provide understanding and help control the dynamics and stabilization of carbohydrate hydrogels by clay minerals.


 Please wait while we load your content...
Please wait while we load your content...