A room-temperature interfacial approach towards iron/nitrogen co-doped fibrous porous carbons as electrocatalysts for the oxygen reduction reaction and Zn–Air batteries†
Abstract
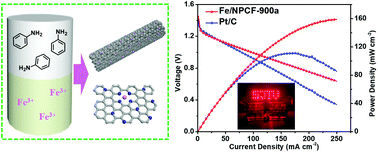
The development of nonprecious and efficient catalysts to boost the oxygen reduction reaction (ORR) is imperative. However, the majority of previously reported approaches suffered from a complicated fabrication procedure, both time consuming and difficult to scale up. Herein, large-scale iron ion embedded polyaniline fibers were successfully fabricated as precursors for preparing iron/nitrogen co-doped fibrous porous carbons (Fe/NPCFs) through an interfacial engineering strategy at room temperature. As ORR electrocatalysts in an alkaline medium (0.1 M KOH), Fe/NPCFs display a positive half-wave potential of 0.827 V (vs. RHE), and high limited current density (up to 5.76 mA cm−2), which are better than those of commercial Pt/C (E1/2 = 0.815 V, JL = 5.47 mA cm−2). Also, Fe/NPCFs exhibit a high ORR catalysis activity (E1/2 = 0.632 V, JL = 5.07 mA cm−2) in acidic medium (0.5 M H2SO4). When used as an air cathode in a primary Zn–air battery, high power density (158.5 mW cm−2) and specific capacity (717.8 mA h g−1) can be easily achieved, outperforming the commercial Pt/C.
- This article is part of the themed collection: Nanocarbons


 Please wait while we load your content...
Please wait while we load your content...