Multifunctional supramolecular self-assembly system for colorimetric detection of Hg2+, Fe3+, Cu2+ and continuous sensing of volatile acids and organic amine gases†
Abstract
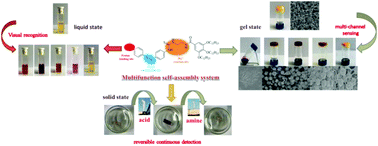
A novel multifunctional gelator (1) based on an azobenzene derivative was designed and characterized. This compound could gelate some solvents including hexane, petroleum ether, DMSO, acetonitrile and ethanol through a heating–cooling procedure. The self-assembly process in different solvents was studied by means of UV-vis absorption and Fourier transform infrared (FTIR) spectra, field emission scanning electron microscopy (FESEM), rheological measurements, X-ray powder diffraction and water contact angle experiments. Interestingly, compound 1 had a high-contrast colorimetric detection ability towards Hg2+, Cu2+, Fe3+ and volatile acids and further organic amine gases in solution through its color change. At the same time, organogel 1 in acetonitrile also exhibited detection performance through a color or gel state change. In the response process, the self-assembly structures were changed from a nanofiber into a microsphere under induction by analytes. More significantly, film 1 could continuously detect volatile acids and organic amine gases. The number of cycles of film 1 for the detection of volatile acids and organic amine gases was at least seven times. The limit of detection (LOD) of film 1 towards TFA was calculated to be 0.0848 ppb. The sensing mechanisms were studied using 1HNMR, FESEM, UV-vis absorption spectra and HRMS. The intramolecular cyclization occurred on molecule 1 and a H2S molecule was lost during the detection process of Hg2+. It was proposed that the –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– bonding could be coordinated by Fe3+ and Cu2+ and this further induced the absorption spectra and color change. For a volatile acid, it was possible that the volatile acid was combined with the N,N-dimethyl amine group of molecule 1. This research opens up a novel pathway to the fabrication of supramolecular self-assembly gels to detect polymetallic ions and trace volatile acids in the environment.
N– bonding could be coordinated by Fe3+ and Cu2+ and this further induced the absorption spectra and color change. For a volatile acid, it was possible that the volatile acid was combined with the N,N-dimethyl amine group of molecule 1. This research opens up a novel pathway to the fabrication of supramolecular self-assembly gels to detect polymetallic ions and trace volatile acids in the environment.


 Please wait while we load your content...
Please wait while we load your content...