3-Layer conductive metal–organic nanosheets as electrocatalysts to enable an ultralow detection limit of H2O2†
Abstract
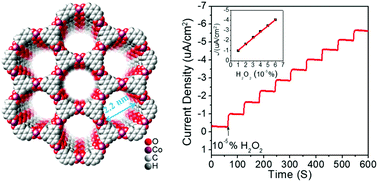
Hydrogen peroxide has been widely studied in cell biology and liquid fuel cells as an oxidant or fuel, and highly efficient and durable electrocatalysts for H2O2 reduction and detection are in high demand. Here, a simple strategy to fabricate conductive 2D single/several-layer [Co3(HHTP)2]n MOF nanosheets, based on 2,3,6,7,10,11-hexahydroxytriphenylene (HHTP) and Co2+ ions, was developed by the Langmuir–Blodgett (LB) method combined with layer-by-layer (LbL) growth technology. The [Co3(HHTP)2]n MOF nanosheets successfully boosted H2O2 reduction with ultrahigh mass activity and good durability, and a new method to detect the H2O2 concentration with an ultralow detection limit of 10−7 (2.9 μmol L−1) was developed. Meanwhile, a series of factors like layer number, surface tension, pH value, ion concentration, and annealing were systematically investigated to further prove the ultrahigh accuracy, sensitivity, and durability of the as-developed H2O2 detection method. The reaction mechanism and energy transfer process of H2O2 reduction catalyzed by the metal–organic nanosheets were investigated by first principles calculations using density functional theory (DFT), showing good agreement with the experiment.


 Please wait while we load your content...
Please wait while we load your content...