Enhancing the photocatalytic properties of PbS QD solids: the ligand exchange approach†
Abstract
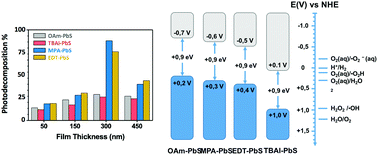
Surface engineering of nanomaterials is a promising tool towards the design of new materials for conversion of solar energy into chemical energy. In this work, we examine the influence of ligand exchange on the photocatalytic performance of solution-processed PbS films. We test different ligands such as oleylamine (OAm), 1,2-ethanedithiol (EDT), 3-mercaptopropionic acid (MPA) and tetrabutylammonium iodide (TBAI). The study demonstrates that PbS films capped with MPA and EDT exhibit 3.5-fold enhanced photocatalytic performance for the photodecomposition of methyl orange upon sunlight exposure. Both band energy alignment and charge carrier transport have a strong impact on the generation of reactive oxygen species (ROS), which play a key role in the photodecomposition process. Moreover, the stability and reusability of the photocatalysts are clearly improved after ligand exchange. We prove how both MPA and EDT provide much more stability to PbS QD films to operate very efficiently up to 8 cycles of photocatalysis. As observed in XPS, the oxidation of PbS is prevented after ligand exchange. We demonstrate how surface chemistry engineering of solution-processed QD films can open a new approach towards the design of highly efficient and stable visible-light-driven photocatalysts, which paves the way to low cost and large area fabrication of high-performance photocatalytic devices.


 Please wait while we load your content...
Please wait while we load your content...