Fe3O4 nanoparticles functionalized with poly(ethylene glycol) for the selective separation and enrichment of Au(iii)
Abstract
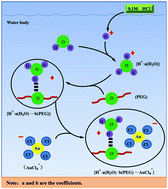
Magnetic nanoparticles containing multiform heteroatoms on their surface have great potential for metal-ion recovery with a high throughput. In this study, a novel adsorbent (Fe3O4@CPTES@PEG) was developed to adsorb Au(III) from aqueous solution, which was synthesized by functionalizing Fe3O4 magnetic nanoparticles with poly(ethylene glycol) via a simple two-step reaction. Various techniques including Fourier transform infrared spectroscopy (FT-IR), thermal gravimetric analysis (TGA), vibrating sample magnetometry (VSM), transmission electron microscopy (TEM) and X-ray diffraction spectroscopy (XRD) were used to characterize the adsorbent, demonstrating that poly(ethylene glycol) was successfully grafted onto the Fe3O4 magnetic nanoparticles. Batch experiments were performed to investigate the effects of the initial concentration of Au(III), contact time and coexisting ions on the absorption of Au(III) from an aqueous solution at pH = 1.0. Simultaneously, the adsorption process was well-fitted with the Langmuir and pseudo-second-order models. The as-synthesized core–shell structured Fe3O4@CPTES@PEG showed a high performance towards Au(III) adsorption with the maximum adsorption capacity of about 83 mg g−1. Significantly, the material exhibited highly selective adsorption toward Au(III) in the presence of coexisting ions (Fe(III), Mn(II), Pb(II), Cd(II), Cu(II), Cr(III), Al(III), Mg(II), Zn(II) and Au(III)). Also, the material could be regenerated using ferrous sulfate, and exhibited excellent reutilization for at least three times without significant loss in its adsorption capacity after a simple post-treatment. Therefore, the proposed method shows enormous potential for practical applications in the selective separation and enrichment of Au(III) from aqueous solution at pH = 1.0.


 Please wait while we load your content...
Please wait while we load your content...