Retracted Article: Development of highly sensitive 1,4-dioxane sensor with semiconductor NiO-doped Nd2O3 nanostructures by electrochemical approach
Abstract
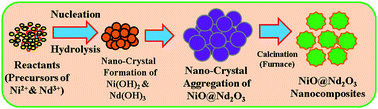
In this approach, 1,4-dioxane, which is an environmental water toxin, is electrochemically identified by using nickel oxide doped on neodymium oxide nanocomposites (NiO@Nd2O3 NCs) embedded on glassy carbon electrode (GCE). Pristine NiO@Nd2O3 NCs were concocted with a simple wet-chemical technique using basic NaOH medium. Nanocomposites were identified with the use of Fourier transform infra-red spectroscopy (FT-IR), ultra-violet visible spectroscopy (UV-Vis), powder X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), field-emission scanning electron microscopy (FESEM), and elemental dispersion spectroscopy (EDS) techniques. 5% Nafion was used as a conducting and coating binder. The application of 1,4-dioxane detection by electrochemical method is mostly based on selectivity, linearity, sensitivity, detection limit (LOD), and limit of detection (LOQ). The plot of calibration curve was linear with linear dynamic range (LDR) of 0.12 mM–0.12 nM. The calculated sensitivity, LOD, and LOQ from the slope were 0.029 μA μM−1 cm−2, 0.033 ± 0.002 nM, and 0.11 ± 0.02 nM respectively. The selective and sensitive chemical sensor fabricated for 1,4-dioxane analyte using doped nanomaterials was simple, cheap, and environment friendly.


 Please wait while we load your content...
Please wait while we load your content...