Catalytic properties of graphene oxide/palladium composites as a function of the fabrication method†
Abstract
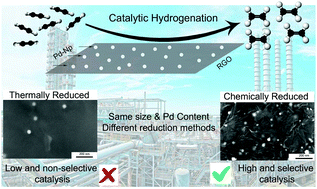
In this work, we used a two-step synthetic procedure to prepare palladium nanoparticles (Pd-NP) on a graphenic support by impregnating graphene oxide (GO) with Pd(II) ions with subsequent thermal or chemical reduction of palladium. The composite materials were prepared with different Pd contents: 1.5% and 15.9%. The intermediate and final products were characterized by TGA, XPS, XRD and SEM. The as-prepared materials were tested for catalysis of the hydrogenation of acetylene to ethylene, which is the critical reaction in the industrial production of polyethylene. The catalysts were tested in a pilot-scale plant reactor at conditions very similar to those used in real industrial processes. The composite material prepared by solid phase thermal reduction exhibited very low catalytic properties due to the formation of the protective graphene layer on the surface of the Pd-NPs; this suggests that the thermal reduction method is not suitable for preparing palladium-based catalysts on carbon supports. The chemically reduced sample with 1.5% Pd content demonstrated good catalytic activity, with conversion factors ranging from 61% through 90% at 40 °C and 90 °C, respectively, and good selectivity. At 15.9% Pd content, the conversion factor was 100% even at 30 °C. The sample performed well even after doubling the flow rate of the feed ethylene mixture compared to the standard rate used for testing commercial catalysts. It successfully functioned for three consecutive days of operation; however, the catalytic activity partially decreased on day 3 due to intensive usage.


 Please wait while we load your content...
Please wait while we load your content...