Surfactant-free fabrication of porous PdSn alloy networks by self-assembly as superior freestanding electrocatalysts for formic acid oxidation†
Abstract
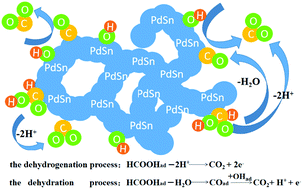
The fine design of three-dimensional (3D) nanocatalysts has attracted great research interest, particularly in fuel cells. In this paper, 3D porous PdSn alloy networks were successfully synthesized for the first time at 60 °C by a facile self-assembly method without using any surfactants. Through comprehensive physical characterization, the formation of a porous network structure with an average diameter of 7 nm was confirmed. The as-prepared PdSn alloy networks exhibited greatly enhanced catalytic performance towards formic acid oxidation (FAO) compared to Pd/C, which could be ascribed to their self-supported network nanostructure, the multifunctional effect between Pd and Sn and the particularly “clean” surface. Also, the widespread amorphous SnO2 greatly enhanced the stability of the network catalysts. Among the three different atomic ratios of PdSn networks, the Pd2Sn1 sample exhibited the highest peak current density (1.35 A mgpd−1), which was 2.3-fold higher than that of Pd/C. After chronoamperometric testing for 3600 s, the Pd2Sn1 networks still showed the highest mass activity (0.16 A mgpd−1), which was 16-fold higher than that of Pd/C. Furthermore, the Pd2Sn1 catalyst displayed the best performance for FAO among all reported PdSn network catalysts. Therefore, after surface electron and geometry modification, the as-obtained PdSn alloy networks can be applied as state-of-the-art electrocatalysts for direct formic acid fuel cells.


 Please wait while we load your content...
Please wait while we load your content...