Boron-decorated C9N4 monolayers as promising metal-free catalysts for electrocatalytic nitrogen reduction reaction: a first-principles study†
Abstract
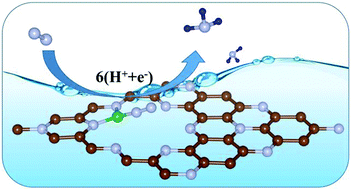
The electrocatalytic N2 reduction reaction (NRR) is a very attractive yet challenging route for sustainable NH3 synthesis under ambient conditions, for which efficient catalysts are required due to the highly strong N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond. In this work, B decorated C9N4 monolayer (B-C9N4) for NRR is studied by using first-principles calculations. It is found that the inert N2 can be sufficiently activated through the electron “acceptance-donation” pattern between N2 and the B dopant. The subsequent N2 reduction prefers the alternating mechanism, and the reduction of *N2 to *N2H is the potential-limiting step with an overpotential of 0.56 V. Furthermore, B-C9N4 exhibits a metallic nature, which can promote charge transfer during the reduction process. The stability of B-C9N4 can be confirmed by the calculated B adsorption energies and the ab initio molecular dynamic simulation. Consequently, owing to its high activity, good electrical conductivity, and excellent stability, B-C9N4 is a promising metal-free N2 reduction catalyst. Our work not only provides an efficient electrocatalyst for NH3 production, but also opens up a promising avenue to develop highly active and robust metal-free catalysts for other applications.
N bond. In this work, B decorated C9N4 monolayer (B-C9N4) for NRR is studied by using first-principles calculations. It is found that the inert N2 can be sufficiently activated through the electron “acceptance-donation” pattern between N2 and the B dopant. The subsequent N2 reduction prefers the alternating mechanism, and the reduction of *N2 to *N2H is the potential-limiting step with an overpotential of 0.56 V. Furthermore, B-C9N4 exhibits a metallic nature, which can promote charge transfer during the reduction process. The stability of B-C9N4 can be confirmed by the calculated B adsorption energies and the ab initio molecular dynamic simulation. Consequently, owing to its high activity, good electrical conductivity, and excellent stability, B-C9N4 is a promising metal-free N2 reduction catalyst. Our work not only provides an efficient electrocatalyst for NH3 production, but also opens up a promising avenue to develop highly active and robust metal-free catalysts for other applications.


 Please wait while we load your content...
Please wait while we load your content...