Phenothiazine electrophores immobilized on periodic mesoporous organosilicas by ion exchange†
Abstract
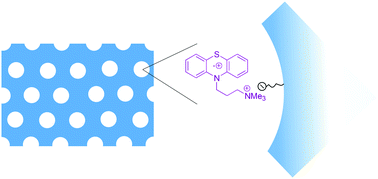
Two different phenothiazines carrying quaternary ammonium groups in the side chain have been synthesized and fully characterized. These compounds were immobilized by ion exchange on the surface of a periodic mesoporous organosilica (PMO) and a neat silica SBA-15 material bearing sulphonate groups covalently bound to the pore surface. The resulting porous and electrochromophoric materials were studied by means of solid state CP-MAS NMR, IR, UV/Vis and fluorescence spectroscopy, nitrogen adsorption/desorption measurements and cyclic voltammetry. Independent of the nature of the support, the colour of these materials changes to pink by irradiation with light, indicating the formation of phenothiazine radical cations. These species, which turned out to be highly stable even in the presence of atmospheric oxygen, were characterized by EPR spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...