Enhanced electrochemical sensing of dopamine in the presence of AA and UA using a curcumin functionalized gold nanoparticle modified electrode†
Abstract
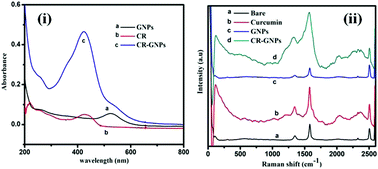
In the present study, a green synthesis of gold nanoparticles was carried out using curcumin as a reducing and functionalizing agent. We have developed a chemically modified electrode using curcumin functionalized gold nanoparticles (CR–GNP) which acted as a redox mediator for the electrochemical oxidation of dopamine in the presence of ascorbic acid and uric acid. Overall, CR–GNP was a stable electrode material and exhibited a synergistic effect towards enhanced electrochemical behavior, increased reaction kinetics, and selectivity in dopamine sensing. Surface characterization, surface composition analysis, physico-chemical property and electrochemical measurements were performed for the CR–GNP modified electrode. This electrode exhibited an enhanced electrochemical behavior towards sensing of dopamine by minimizing the oxidation potential from +0.23 V to +0.18 V and the CR–GNP modified electrode showed a higher oxidation peak current when compared with the bare and curcumin modified electrodes. Hydrodynamic measurements of dopamine were carried out using the chronoamperometry technique and static measurements were carried out using cyclic voltammetry and linear sweep voltammetry techniques. Under optimal conditions the modified electrode exhibited linearity in the calibration plot for dopamine concentrations ranging from 0.4 × 10−6 to 5.6 × 10−5 M. This work explored selectivity towards oxidation of dopamine in the presence ascorbic acid and uric acid with a detection limit of 4.2 × 10−8 M.


 Please wait while we load your content...
Please wait while we load your content...