The necessity of free and uncrowded coordination environments in biomimetic complex models: oxidative coupling by mixed-ligand cobalt(ii) complexes of diazene–disulfonamide†
Abstract
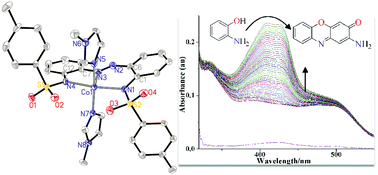
The importance of phenoxazinone chromophore has inspired chemists to develop new synthetic models that can mimic phenoxazinone synthase activity. However, a systematic approach for the investigation of structure–property correlation of model complexes based on the appreciable number of structurally varied and well-defined complexes is scarce. In this study, two new diazene–disulfonamide ligands RSO2–NH–Ph–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–Ph–NH–SO2R (where R = methyl (1) and R = tolyl (2)) were deployed along with monodentate N-donors derived from imidazole or pyridyl moieties to assemble mixed-ligand cobalt(II) complexes Co1(im)2, Co1(py)2, Co1(dpy)2, Co2(im)2 and Co2(py)2. Based on the structural characterization results, the diazene–disulfonamide ligands coordinated as tridentate dianionic N^N^N-donors, while two units of the monodentate N-donors aided to complete a square-pyramidal geometry around each cobalt(II) metal centres. Continuous shape measurements (CShM) calculations were carried out to further clarify the distorted square-pyramidal geometry as a vacant octahedral vOC-5 (C4v) coordination environment. A comparative study of the phenoxazinone synthase mimicking behaviour of the complexes was conducted and the results led to conclusions that the transition cobalt(II) active sites performed better when uncrowded and in the presence of readily detachable co-ligands. Consequently, from the experimental point of view, the remarkable reactivity of metallo-enzymes was pictured to depend on the availability of vacant metal coordination positions that are not sterically clogged.
N–Ph–NH–SO2R (where R = methyl (1) and R = tolyl (2)) were deployed along with monodentate N-donors derived from imidazole or pyridyl moieties to assemble mixed-ligand cobalt(II) complexes Co1(im)2, Co1(py)2, Co1(dpy)2, Co2(im)2 and Co2(py)2. Based on the structural characterization results, the diazene–disulfonamide ligands coordinated as tridentate dianionic N^N^N-donors, while two units of the monodentate N-donors aided to complete a square-pyramidal geometry around each cobalt(II) metal centres. Continuous shape measurements (CShM) calculations were carried out to further clarify the distorted square-pyramidal geometry as a vacant octahedral vOC-5 (C4v) coordination environment. A comparative study of the phenoxazinone synthase mimicking behaviour of the complexes was conducted and the results led to conclusions that the transition cobalt(II) active sites performed better when uncrowded and in the presence of readily detachable co-ligands. Consequently, from the experimental point of view, the remarkable reactivity of metallo-enzymes was pictured to depend on the availability of vacant metal coordination positions that are not sterically clogged.


 Please wait while we load your content...
Please wait while we load your content...