Self-assembly and photo-responsive behavior of bis-terpyridyl Eu3+-complex L1†
Abstract
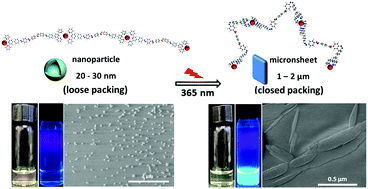
In the present paper, a bis-terpyridyl Eu3+ complex (bis-terpyridyl Eu3+-complex L1) was synthesized through the coordination between Eu3+ and a compound L1 with an azobenzene-functionalized chain between the two terpyridine ligands. The assembly behavior of bis-terpyridyl Eu3+-complex L1, which could be an excellent ultraviolet-responsive luminescent material, was investigated. We found that the introduction of an azobenzene-functionalized chain between bis-terpyridine ligands enabled the Eu3+ complex to undergo photo-isomerization and control the aggregation behavior in a solution. Upon irradiation by 365 nm ultraviolet light, the methanol solution of the Eu3+complex exhibited visible fluorescence enhancement with an order of magnitude improvement in the fluorescence quantum yield and fluorescent lifetime. In particular, the photo-induced isomerization of azobenzene induced the transition of aggregates from nanospheres to micron sheets. The transition of aggregates was demonstrated by dynamic laser light scattering (DLS) data, atomic force microscopy (AFM) observations and Gaussian simulations. After UV irradiation, the tight accumulation of fluorophores in bis-terpyridyl Eu3+-complex L1 was speculated to be the dominant factor of the enhanced photoluminescence property. Our study may help expand the research on azobenzene-functionalized molecules based on coordination and provide a promising way for preparing responsive fluorescent materials.


 Please wait while we load your content...
Please wait while we load your content...