Efficient removal of low-concentration Cr(vi) from aqueous solution by 4A/HACC particles
Abstract
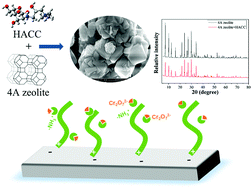
A new cationic surface-modified 4A zeolite for adsorbing trace chromium in aqueous solution was successfully synthesized. Detailed structural characterization and adsorption mechanism analysis were carried out by zeta potential measurements, SEM, XRD and XPS. The results showed that surface modification did not destroy the excellent pore structure of 4A zeolite itself and the surface roughness leads to high hydrophilicity. HACC was evenly distributed on the surface of 4A zeolite, each amino group site on HACC having adsorption properties. The results of sorption behavior showed that the adsorption efficiency of the modified adsorbent for low-concentration Cr(VI) reached up to 92%, which was significantly higher than that of each component alone. The thermodynamic and kinetic parameters demonstrated the removal process to be spontaneous, exothermic and pseudo-second-order chemisorption. Moreover, the adsorption performance is little affected by pH.


 Please wait while we load your content...
Please wait while we load your content...