A newly designed isoindigo/thiophene medium-sized molecule containing a π (D–A–D) bridge with unexpected organic photovoltaic performance†
Abstract
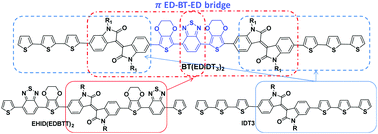
4,7-Bis(2,3-dihydrothieno[3,4-b][1,4]dioxin-5-yl)benzo[c][1,2,5]thiadiazole (ED-BT-ED) is used as a π (donor–acceptor–donor) bridge between two arms of (E)-1-(2-octyldodecyl)-3-(1-(2-octyldodecyl)-2-oxo-6-(5-(5-(thiophen-2-yl)thiophen-2-yl)thiophen-2-yl)indolin-3-ylidene)indolin-2-one (ID-T3) to form a novel medium-sized molecule BT(EDIDT3)2, using a Pd(AcO)2 catalyzed direct C–H arylation method. The presence of a π(D–A–D) bridge induced π conjugation extension, across the molecule backbone structure, the octyldodecyl groups improved the solubility and the terminal thiophene units enhanced the absorption in the UV-vis region. The optical and electrochemical properties of BT(EDIDT3)2 were studied. The BT(EDIDT3)2 : PC70BP (1 : 1) blend was prescreened as an active layer for organic solar cells and showed a power conversion efficiency (PCE)% = 2.25%, as an unexpected result compared with its counterparts with shorter conjugation lengths. The unexpected photovoltaic performance of BT(EDIDT3)2 highlights the importance of the careful selection of the inserted π-aromatic groups because their proper choice is essential to enhance the light-harvesting efficiency. Calculations by density functional theory (DFT) and time-dependent density functional theory (TD-DFT) were performed to confirm this deduction. We deduced that the central benzothiadiazole core in BT(EDIDT3)2 made big distortions that could inhibit the benefit behind the extension of conjugated molecular length.


 Please wait while we load your content...
Please wait while we load your content...