Fluorinated boron subphthalocyanines: Lewis acid based templating chemistry facilitates random halide exchange, and fluoride versus chloride affects the basic photophysical properties and the solid-state arrangement†
Abstract
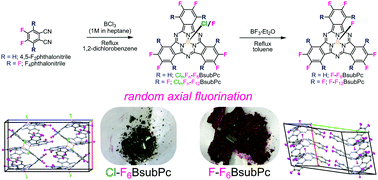
A sub-group of the phthalocyanine family, the boron subphthalocyanines (BsubPcs), have robust chemistry and can be readily modified at the axial and peripheral positions to tune their physical properties. Our laboratory recently discovered that peripheral chlorination is highly influential on the physical properties of BsubPcs and their application into organic electronic devices. Owing to this and for this study, we have now investigated the effect of peripheral and axial fluorination versus chlorination. In order to obtain the BsubPcs for these points of comparisons, we first studied and optimized their synthetic procedure. This was needed as it was found that the process of reacting the Lewis acid BCl3 with a (fluoro)n-phthalonitrile yielded a mixture of Cl-FnBsubPc and F-FnBsubPc with varying compositions on each experimental run; the axial fluoride compound being formed through an unexpected fluoride exchange. High purity axial fluoride BsubPcs were then achieved via completion of the fluoride exchange using BF3·OEt2, which required further process consideration. Final purification was completed by train sublimation. The high purity obtained for F-F6BsubPc and F-F12BsubPc enabled direct points of comparisons against chlorinated analogs to understand the role of relative halogenation in the periphery and axial positions of BsubPcs on their photophysical properties. We also report for the first time single crystal structures of Cl-F6BsubPc and F-F6BsubPc and confirm previously published crystal structures of Cl-F12BsubPc and F-F12BsubPc. It is clear that the axial fluoride significantly influences the basic solid-state arrangement.


 Please wait while we load your content...
Please wait while we load your content...