PAMAM dendrimers with a porphyrin core as highly selective binders of Li+ in an alkaline mixture. A spectroscopic study
Abstract
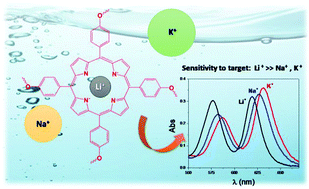
The interactions between a series of dendrimers with a porphyrin core (Pf-Ds) and alkaline cations (Li+, Na+ and K+) in DMF solution were studied by absorption and fluorescence spectroscopies. The solutions basified with an excess of NaOH or KOH showed spectral shifts that could be ascribed to the complexes formed between the Pf-Ds anions and the cations. Subsequent spectral shifts were observed by adding an aqueous solution of LiCl to the basic Pf-Ds solutions, at lithium concentrations several orders of magnitude lower than those of Na+ or K+. This striking preference towards Li+ was interpreted based on the formation of a more stable complex, since the smallest size of this ion fits in the deprotonated porphyrin core. Linear increases in the fluorescence emission were detected with sub-micromolar lithium concentrations, for all Pf-Ds studied. One of the compounds, namely C9, showed a linear fluorescence response at lithium concentrations as low as a few tens of nanomoles per liter. Both the selectivity and sensitivity observed suggest that the Pf-Ds analyzed have potential to be applied in sensing of Li+ in solution.


 Please wait while we load your content...
Please wait while we load your content...