Preparation of a PdRuNi/C tri-metallic nanocatalyst and its excellent catalytic performance for ethylbenzene hydrogenation reaction†
Abstract
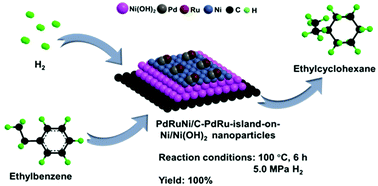
Herein, Ni/C was prepared at room temperature (RT) by liquid chemical reduction by using hydrazine hydrate as the reducing agent. The impregnation method was adopted to deposit precious metals on carbon black (C) to obtain the Pd/C, Ru/C monometallic and PdRu/C bimetallic catalysts. The PdNi/C, RuNi/C bimetallic and PdRuNi/C tri-metallic catalysts were obtained through a chemical replacement reaction method. The as-prepared catalysts were characterized by inductively coupled plasma mass spectrometry (ICP-MS), X-ray diffraction (XRD), transmission electron microscopy (TEM), high resolution TEM (HRTEM), high-angle annular dark-field scanning TEM (HAADF-STEM) and STEM energy dispersive X-ray spectroscopy (STEM-EDS) elemental mapping and line-scanning, scanning electron microscopy (SEM) and SEM-EDS, X-ray photoelectron spectroscopy (XPS) and high-sensitivity low-energy ion scattering spectroscopy (HS-LEIS) techniques, which proved that PdRu islands were supported on the Ni/Ni(OH)2 nanoparticles (NPs) (PdRu-on-Ni/Ni(OH)2 NPs) in PdRuNi/C. The results revealed that the PdRuNi/C catalyst showed the most excellent catalytic performance for ethylbenzene hydrogenation under mild reaction conditions mainly owing to the synergistic effect among the Pd, Ru and Ni related species. The PdRuNi/C catalyst also exhibited good stability in ethylbenzene hydrogenation.


 Please wait while we load your content...
Please wait while we load your content...