Does poly(ionic liquid) modulate the non-covalent interactions of chicken egg white lysozyme? Elucidation of biomolecular interactions between biomolecules and macromolecular solvents†
Abstract
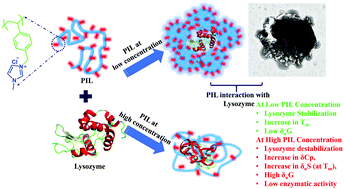
Poly(ionic liquids) (PILs) have recently generated much interest in the material chemistry field because of their wide range of applications. However, the use of PILs as a co-solvent for protein stability is relatively still unknown. Therefore, in this work, we report the synthesis and characterization of vinylbenzyl chloride and N-methylimidazole-based PILs and their biomolecular interactions with the antimicrobial enzyme lysozyme. Different spectroscopic, thermal, and morphological studies indicated that PILs at lower concentrations (0.005–0.2 mg mL−1) do not affect the structural or thermal (only 0.005 mg mL−1) stability of lysozyme. However, the functional stability, as determined by enzymatic activity of the lysozyme, decreased with increasing concentration of PIL. Furthermore, higher Tm values do not necessarily indicate higher thermal stability of the protein. Alternatively, a combination of a complete set of thermodynamic parameters such as Tm, δCp and δuS (at Tm), and δuG is more helpful in predicting protein stability. Morphological changes observed using TEM also showed the interaction of the PIL with lysozyme at lower concentrations, whereas the protein denatured at higher concentrations of PIL. This is the first stability study using PIL as a co-solvent for lysozyme, and it paves the way for future similar studies using different classes of PILs to evaluate protein stability.


 Please wait while we load your content...
Please wait while we load your content...