Synthesis and comparison of copper(ii) complexes with various N-aminotetrazole ligands involving trinitrophenol anions†‡
Abstract
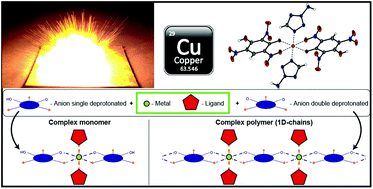
Due to the ongoing research on lead-free energetic materials, two different ligand systems (1-amino-5H-tetrazole (1-AT), 2-amino-5H-tetrazole (2-AT), 1-amino-5-methyltetrazole (1-AMT), and 2-amino-5-methyltetrazole (2-AMT)) were applied for the synthesis of 12 new energetic coordination compounds (ECC) with copper(II) as the central metal. Different anions based on trinitrophenols (picric acid (HPA), styphnic acid (H2TNR), and trinitrophloroglucinol (H3TNPG)) were used for the specifical tuning of the energetic and physico-chemical properties of the complexes. Through the choice of ligand, the characteristics of the resulting products can be easily adjusted either towards sensitive primary explosives usable for classical initiation setups or towards laser-ignitable explosives with decreased sensitivities. The ECC were extensively characterized by e.g., X-ray diffraction (XRD), elemental analysis (EA), IR, differential thermal analysis (DTA), and UV/vis. In addition, the most promising compounds were analyzed by TGA and in classical initiation tests using nitropenta (PETN). Furthermore, the sensitivities towards external stimuli (impact, friction, and electrostatic discharge) were determined by standard methods and the influence of the anions towards them was investigated. Compounds [Cu(TNR)(1-AMT)2] and [Cu(HTNPG)(1-AMT)2] both possess appropriate sensitivities as well as thermal stabilities above 200 °C and show promising results to be used as potential lead azide replacements. In addition, all ECC were irradiated with a near infrared light (NIR) laser diode leading to different responses.


 Please wait while we load your content...
Please wait while we load your content...