Urea-modified grass ash activated sludge carbon: structure and adsorption properties towards H2S and CH3SH
Abstract
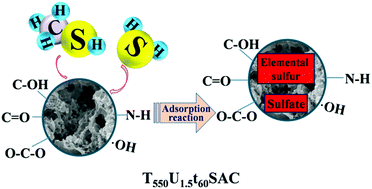
The sludge from a sewage treatment plant was used as the raw material, the plant ash was used as the activator, urea was used as the modifier and the sludge was used to prepare the sludge carbon material. Orthogonal tests were designed to determine the best preparation conditions for the samples. The structure and deodorization products of sludge carbon were analyzed by SEM, FT-IR, XPS, specific surface area and pore structure analyses. The results showed that the sludge carbon T550U1.5t60SAC had the best deodorization performance when the pyrolysis temperature was 550 °C, the stucco/drug ratio was 1 : 1.5, and the pyrolysis time was 60 min under N2 atmosphere. The penetration time for hydrogen sulfide and methyl mercaptan reached 200 min and 100 min, respectively. Structural analysis showed that the T550U1.5t60SAC carbon material had a rough surface and wide pore diameter distribution. It not only had oxygen-containing functional groups on the surface, but the concentration of the nitrogen-containing functional groups on the surface also increased with the addition of urea, and the surface alkalinity of the material increased. These factors provided channels and activity points for the adsorption of H2S and CH3SH. The surface area and pore size distribution of the adsorbed T550U1.5t60SAC sludge changed significantly, particularly after the micropores disappeared. This indicated that the deodorization reaction mainly occurred in the micropores, and deodorized products such as elemental sulfur and sulfate appeared. The analysis suggested that the ˙OH radical on the surface of sludge carbon was the main species for the oxidation of H2S and CH3SH.


 Please wait while we load your content...
Please wait while we load your content...